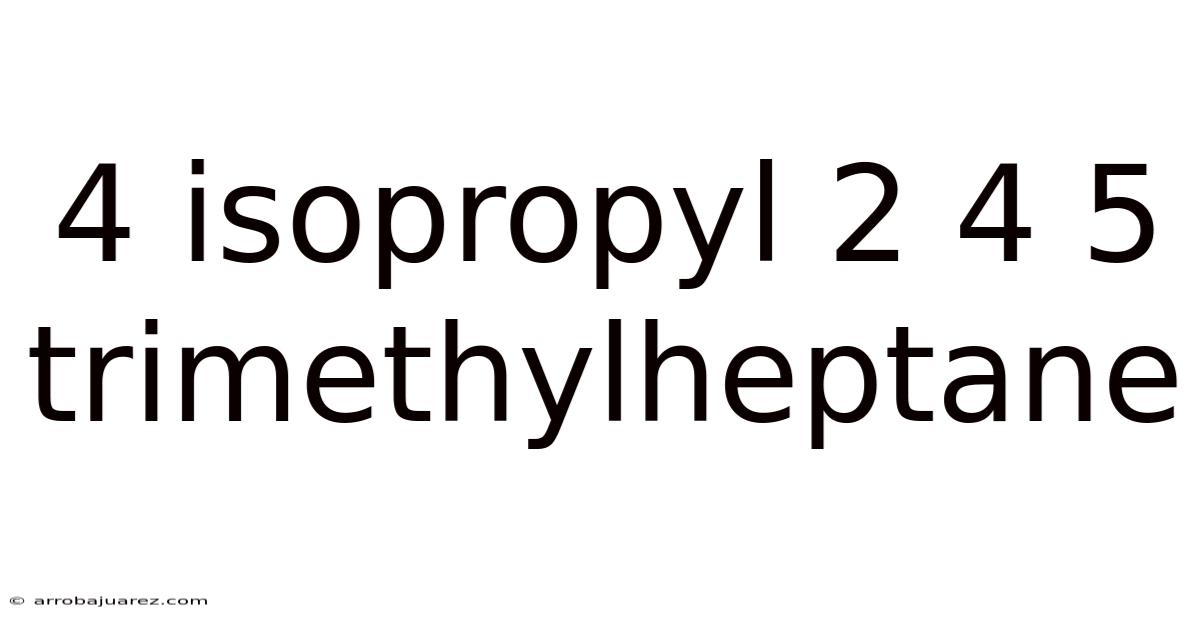Unveiling the Secrets of 4-Isopropyl-2,4,5-Trimethylheptane: A Deep Dive
4-Isopropyl-2,4,5-trimethylheptane, a complex and fascinating organic compound, belongs to the alkane family. Its unique structure and properties have garnered attention in various scientific fields. This article breaks down the intricacies of this molecule, exploring its structure, synthesis, properties, applications, and potential impact.
Understanding the Molecular Architecture
The name itself provides a roadmap to understanding the structure of 4-isopropyl-2,4,5-trimethylheptane. Let's break it down:
- Heptane: This indicates the presence of a seven-carbon straight chain as the backbone of the molecule.
- 2,4,5-Trimethyl: This signifies three methyl groups (CH3) attached to the second, fourth, and fifth carbon atoms of the heptane chain.
- 4-Isopropyl: This indicates an isopropyl group (CH(CH3)2) attached to the fourth carbon atom of the heptane chain.
Which means, the molecule consists of a seven-carbon chain with three methyl groups and one isopropyl group branching off at specific locations. In practice, this highly branched structure significantly influences its physical and chemical properties. The systematic IUPAC name of this compound is 4-(propan-2-yl)-2,4,5-trimethylheptane.
Synthesizing 4-Isopropyl-2,4,5-Trimethylheptane
The synthesis of such a complex branched alkane typically involves multi-step organic reactions. Direct synthesis from simpler starting materials can be challenging due to the need for precise control over the placement of each substituent. Here are a few potential synthetic routes:
-
Grignard Reaction Approach: This method could involve reacting a Grignard reagent with a suitable carbonyl compound to introduce the isopropyl group. Subsequently, methylation reactions can be employed to add the methyl groups at the desired positions. Protecting group chemistry might be necessary to prevent unwanted side reactions And that's really what it comes down to..
- Step 1: Formation of Grignard Reagent: Isopropyl magnesium halide (e.g., isopropylmagnesium bromide) can be prepared by reacting isopropyl halide with magnesium metal in an ethereal solvent like diethyl ether or tetrahydrofuran (THF).
- Step 2: Reaction with Carbonyl Compound: The Grignard reagent reacts with a suitable carbonyl compound (e.g., a ketone or aldehyde) that has the appropriate carbon chain length and protecting groups to control the subsequent methylation steps.
- Step 3: Methylation Reactions: Methyl groups can be introduced using methylation reagents like methyl iodide (CH3I) or dimethyl sulfate ((CH3)2SO4) in the presence of a strong base. Protecting groups would be crucial to direct the methylation to the desired carbon atoms (C-2, C-4, and C-5).
- Step 4: Deprotection: After all the substituents are in place, the protecting groups are removed to yield the target molecule.
-
Alkylation Reactions: Another approach could involve sequential alkylation reactions using alkyl halides and strong bases. Again, careful selection of protecting groups and reaction conditions is crucial to ensure the correct regiochemistry.
- Step 1: Formation of Carbanion: A heptane derivative with appropriate protecting groups is treated with a strong base (e.g., lithium diisopropylamide - LDA) to form a carbanion at the desired carbon atom.
- Step 2: Alkylation with Alkyl Halide: The carbanion reacts with an alkyl halide (e.g., methyl iodide or isopropyl bromide) to introduce the alkyl group.
- Step 3: Repetition of Alkylation: The process is repeated with different alkyl halides and protecting groups to introduce the remaining methyl and isopropyl groups at the correct positions.
- Step 4: Deprotection: Finally, the protecting groups are removed to obtain the final product.
-
Multi-Component Coupling Reactions: Modern synthetic methodologies, such as transition-metal catalyzed coupling reactions, could also be employed to assemble the molecule from smaller fragments. These reactions often offer high selectivity and efficiency Practical, not theoretical..
- This advanced approach would likely involve the synthesis of several key building blocks, each containing a portion of the final molecule with the desired substituents. These building blocks would then be coupled together using sophisticated catalytic reactions. While complex, this strategy can offer higher yields and better control over stereochemistry compared to traditional methods.
Challenges in Synthesis:
The synthesis of 4-isopropyl-2,4,5-trimethylheptane presents several challenges:
- Stereocontrol: The molecule has chiral centers (C-2, C-4, and C-5), leading to the possibility of multiple stereoisomers. Achieving stereoselective synthesis, where only one specific stereoisomer is formed, is a significant challenge.
- Regioselectivity: Introducing the alkyl groups at the correct positions on the heptane chain requires careful control of regioselectivity. Protecting groups and specific reaction conditions are essential to avoid unwanted side reactions.
- Yield: Multi-step syntheses often suffer from low overall yields due to losses in each step. Optimizing each reaction step to maximize yield is crucial for a successful synthesis.
Due to these complexities, the synthesis of 4-isopropyl-2,4,5-trimethylheptane is not a common laboratory procedure and would likely be undertaken only for specific research purposes Not complicated — just consistent..
Exploring the Physical and Chemical Properties
The highly branched structure of 4-isopropyl-2,4,5-trimethylheptane dictates its unique physical and chemical properties.
- Physical State: At room temperature, it is expected to be a colorless liquid. The branching hinders efficient packing of the molecules, leading to a lower melting point and boiling point compared to its straight-chain isomer.
- Boiling Point: Due to the branching, the boiling point is expected to be relatively low compared to a straight-chain alkane with the same number of carbon atoms. The branched structure reduces intermolecular forces (van der Waals forces), making it easier for the molecules to escape into the gas phase.
- Density: The density is also expected to be lower than that of a straight-chain alkane. Branching increases the volume occupied by each molecule, thus decreasing the density.
- Solubility: As an alkane, it is highly nonpolar and thus insoluble in water. It is, however, soluble in other nonpolar organic solvents like hexane, benzene, and diethyl ether.
- Chemical Inertness: Alkanes are generally known for their low reactivity. 4-Isopropyl-2,4,5-trimethylheptane is relatively unreactive towards most common reagents under normal conditions. On the flip side, it can undergo combustion in the presence of oxygen at high temperatures.
- Spectroscopic Properties: Techniques like Nuclear Magnetic Resonance (NMR) spectroscopy and Mass Spectrometry (MS) can be used to characterize the molecule. NMR spectroscopy provides detailed information about the connectivity of atoms and the chemical environment of each hydrogen and carbon atom. Mass spectrometry provides information about the molecular weight and fragmentation pattern of the molecule.
Predicted Properties (Based on Analogy to Similar Compounds):
it helps to note that specific physical and chemical properties would need to be experimentally determined. Still, based on the properties of similar branched alkanes, we can predict some characteristics:
- Viscosity: Likely to have a relatively low viscosity due to the branching, which hinders intermolecular interactions.
- Refractive Index: Expected to have a characteristic refractive index, which can be used for identification and purity assessment.
- Flash Point: Will have a flash point, which is the lowest temperature at which it can form an ignitable mixture in air. This is important for safety considerations during handling and storage.
Potential Applications and Research Areas
While 4-isopropyl-2,4,5-trimethylheptane may not have widespread industrial applications, its unique structure makes it a subject of interest in certain research areas:
-
Fuel Chemistry: Branched alkanes are often used as components in gasoline to improve its octane rating. The highly branched structure of 4-isopropyl-2,4,5-trimethylheptane could potentially contribute to improved combustion properties and reduced engine knocking. Further research would be needed to determine its specific performance characteristics.
-
Lubricant Industry: Synthetic lubricants often contain branched alkanes to enhance their viscosity and thermal stability. The molecule's structure might offer advantages in specific lubricant formulations, especially in applications requiring low-temperature performance Simple as that..
-
Polymer Science: Alkanes can be used as solvents or as building blocks in the synthesis of certain polymers. 4-Isopropyl-2,4,5-trimethylheptane could potentially be incorporated into polymer chains to modify their properties, such as flexibility or hydrophobicity.
-
Fundamental Research: The molecule serves as an excellent model compound for studying the relationship between molecular structure and physical properties. Researchers can use it to test theoretical models and computational methods for predicting the behavior of complex molecules. Its synthesis and characterization can also advance the development of new synthetic methodologies That's the part that actually makes a difference..
-
Chemical Sensors: Functionalized derivatives of this alkane could potentially be used in chemical sensors. By attaching specific functional groups that interact with target analytes, the molecule could be used to detect the presence of specific substances in a sample Practical, not theoretical..
Future Research Directions:
- Detailed Spectroscopic Analysis: A comprehensive spectroscopic analysis, including NMR, IR, and Raman spectroscopy, would provide valuable information about the molecule's structure and vibrational modes.
- Computational Studies: Computational chemistry methods can be used to predict its properties, such as boiling point, density, and heat of formation. These calculations can complement experimental measurements and provide insights into the molecule's behavior.
- Structure-Property Relationship Studies: Investigating the impact of branching on the physical properties of alkanes is an ongoing area of research. 4-Isopropyl-2,4,5-trimethylheptane can serve as a valuable data point in these studies.
Safety Considerations and Handling
As with any chemical compound, proper safety precautions should be taken when handling 4-isopropyl-2,4,5-trimethylheptane The details matter here. That's the whole idea..
- Flammability: Alkanes are flammable, so it should be kept away from open flames and other sources of ignition.
- Inhalation and Skin Contact: Inhalation of vapors and skin contact should be avoided. Proper ventilation and the use of gloves and eye protection are recommended.
- Storage: It should be stored in a tightly closed container in a cool, dry, and well-ventilated area.
- Disposal: Waste should be disposed of in accordance with local regulations.
Disclaimer: The information provided in this article is for educational purposes only and should not be considered as professional advice. Always consult with qualified professionals for specific applications and safety information The details matter here..
Frequently Asked Questions (FAQ)
-
What is the IUPAC name of 4-isopropyl-2,4,5-trimethylheptane?
The IUPAC name is 4-(propan-2-yl)-2,4,5-trimethylheptane.
-
**Is 4-isopropyl-2,4,5-trimethylheptane a polar or nonpolar molecule?
It is a nonpolar molecule due to its alkane structure, consisting primarily of carbon and hydrogen atoms.
-
**What are the potential applications of this compound?
Potential applications include fuel chemistry, lubricant industry, polymer science, and fundamental research Worth knowing..
-
**Is the synthesis of 4-isopropyl-2,4,5-trimethylheptane straightforward?
No, the synthesis is complex due to the need for precise control over regioselectivity and stereoselectivity. Because of that, protecting group chemistry is often required. 5. **What safety precautions should be taken when handling this compound?
It should be handled with care, avoiding inhalation, skin contact, and exposure to open flames. That said, proper ventilation, gloves, and eye protection are recommended. 6. **What kind of spectroscopic techniques can be used to characterize it?
NMR spectroscopy and mass spectrometry are useful techniques for characterization Simple as that..
-
**What are the chiral centers in the molecule?
Carbon atoms C-2, C-4, and C-5 are chiral centers in the molecule That's the whole idea..
-
**Is this compound naturally occurring?
No, this compound is not typically found in nature and must be synthesized. Here's the thing — 9. **Does branching affect the boiling point of alkanes?
Yes, branching generally lowers the boiling point of alkanes compared to their straight-chain isomers.
-
**What are the main challenges in synthesizing highly branched alkanes like this one?
The main challenges include controlling regioselectivity (where substituents attach) and stereoselectivity (the spatial arrangement of atoms) during the synthesis And it works..
Conclusion: A Complex Molecule with Intriguing Potential
4-Isopropyl-2,4,5-trimethylheptane, a highly branched alkane, presents a fascinating case study in organic chemistry. While its synthesis is challenging, the potential applications in fuel chemistry, lubricant industry, and polymer science warrant further investigation. Practically speaking, its unique structure dictates its physical and chemical properties, making it a subject of interest in various research areas. The study of such complex molecules not only enhances our understanding of structure-property relationships but also drives innovation in chemical synthesis and materials science. So as synthetic methodologies continue to advance, more efficient routes to synthesize this and similar complex molecules may emerge, unlocking their full potential in various scientific and industrial applications. Further research into this compound could reveal novel applications and contribute to advancements in various fields And it works..
Easier said than done, but still worth knowing.